Arsenic is the 53rd most abundant element in the Earth’s crust, comprising about 1.5 parts per million (0.00015%). [citation needed] Typical background concentrations of arsenic do not exceed 3 ng/m 3 in the atmosphere; 100 mg/kg in soil; 400 μg/kg in vegetation; 10 μg/L in freshwater and 1.5 μg/L in seawater.. The number of protons determines the element, but the number of neutrons in the atom of any one element can vary. Each variation is an isotope. About 14 radioactive isotopes of arsenic are known also.

Arsenic by Destiny French

Arsenic Atomic Number Atomic Mass Density of Arsenic

Arsenic Electron configuration Symbol Atomic Number Atomic Mass Oxidation States

Electronic Structure Of Arsenic , Free Transparent Clipart ClipartKey

The Periodic Table Chemistry for Majors

Periodic Table Atomic Number 33 Periodic Table Timeline

A Molecule With 14 Total Electrons and 12 Total Protons
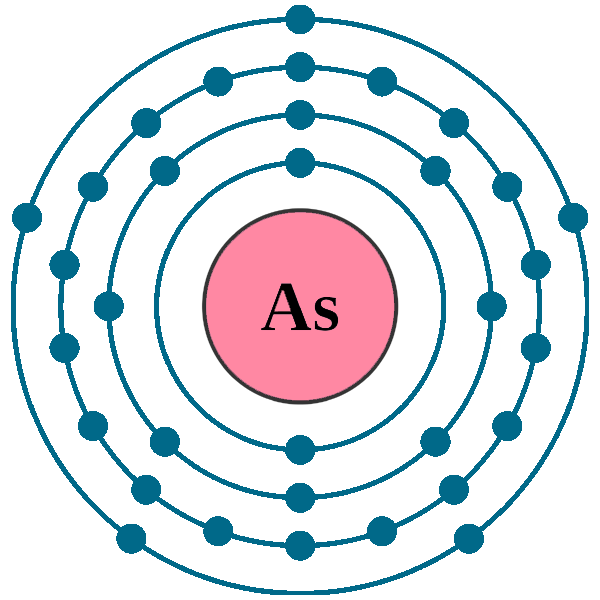
Arsenic As (Element 33) of Periodic Table Elements FlashCards

Atomic Number Arsenic stock illustration. Illustration of configuration 55455396
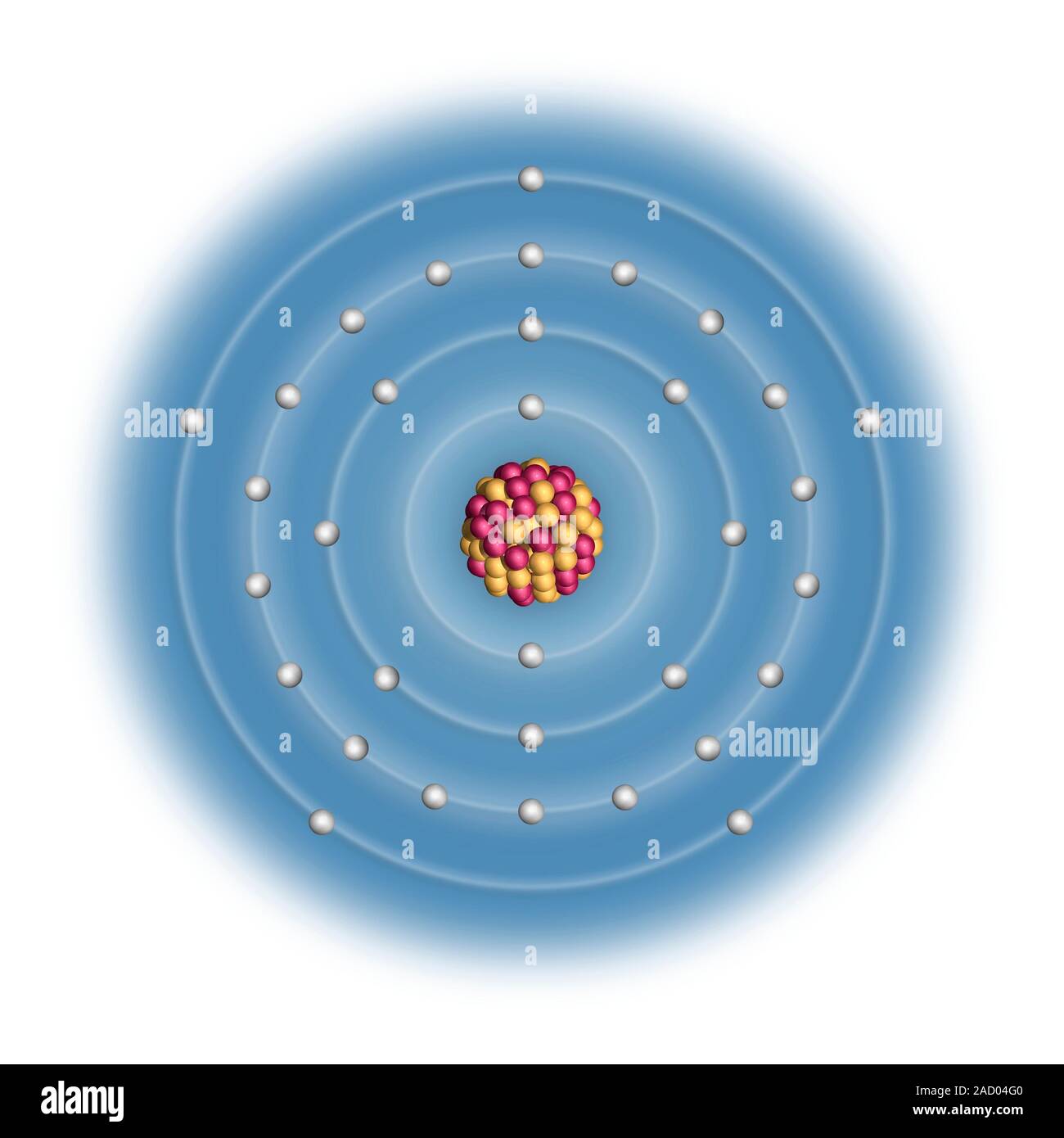
Arsenic (As). Diagram of the nuclear composition and electron configuration of an atom of

SOLVEDUsing the Periodic Table determine the number of protons in an atom of each of the

Protons, Neutrons, Electrons for Arsenic (As, As3)

How To Find Protons On Periodic Table

how many protons and electrons in Arsenic ion

Arsenic Element Properties and Information Chemical Engineering World

Arsenic, atomic structure Stock Image C018/3714 Science Photo Library Atomic structure
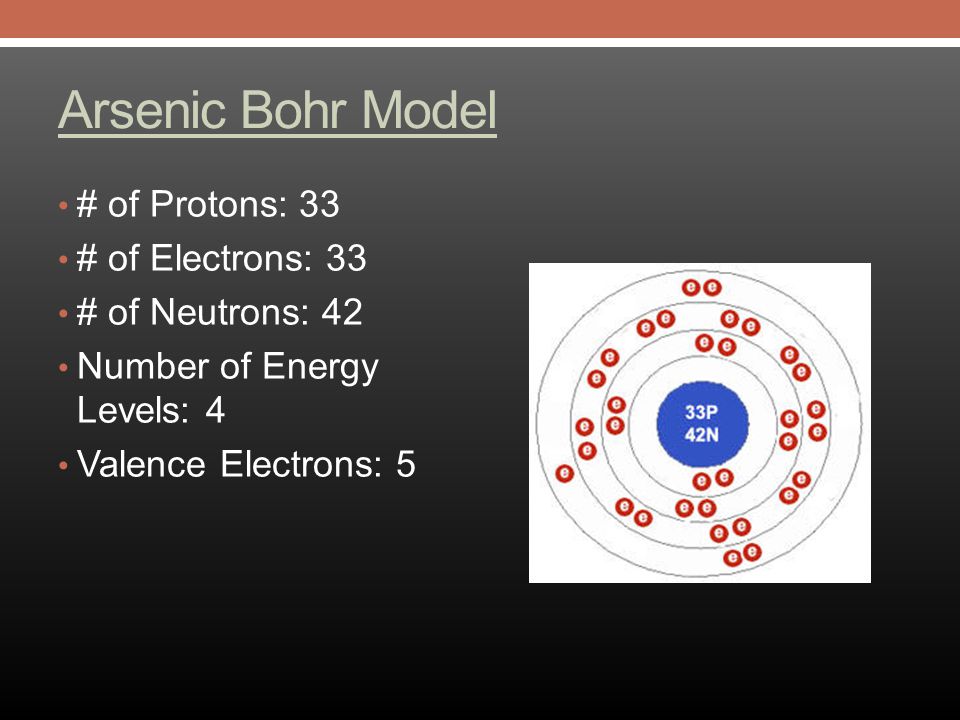
Arsenic Electron Configuration (As) with Orbital Diagram
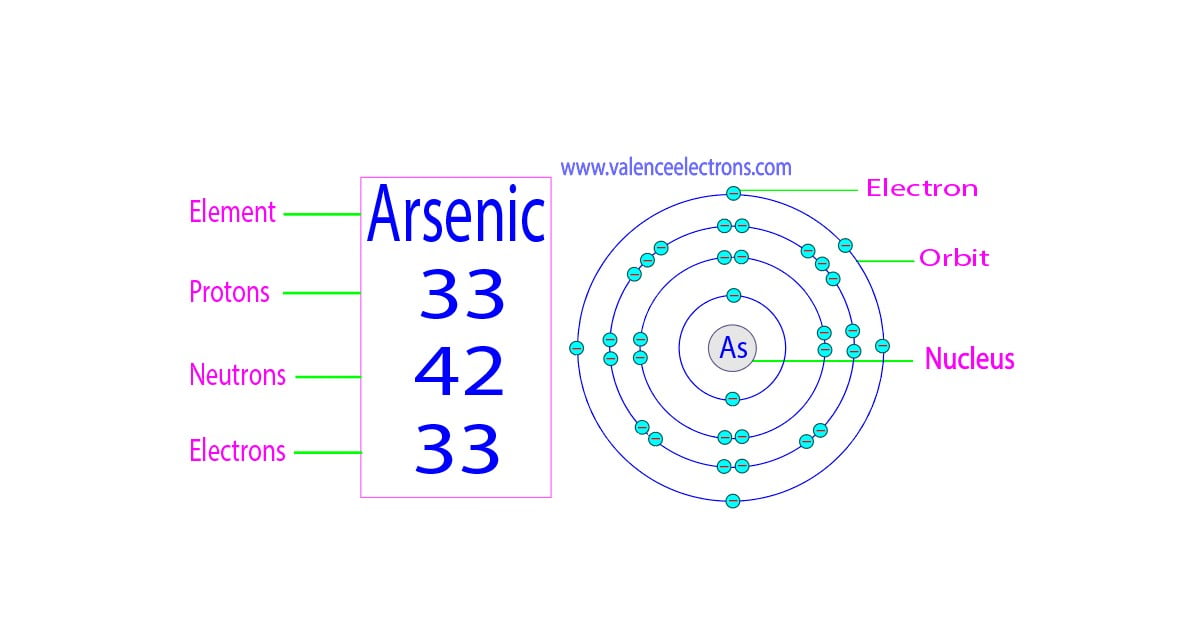
Protons, Neutrons, Electrons for Arsenic (As, As3)

Arsenic, atomic structure Stock Image C013/1577 Science Photo Library
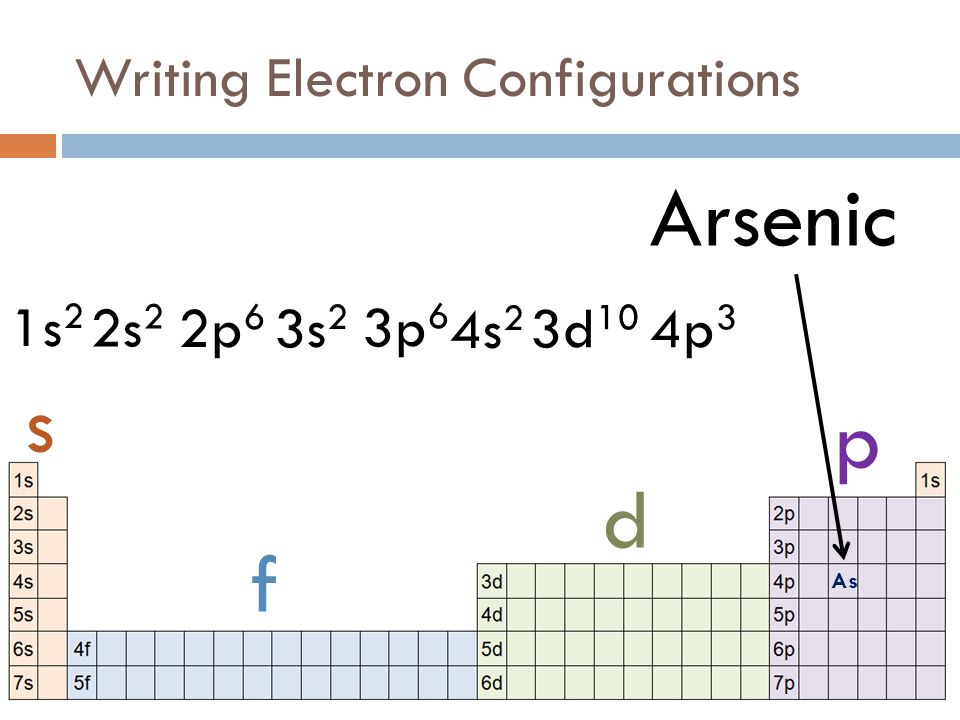
How To Find A Electron Configuration For Arsenic
Protons: 33: Neutrons in most abundant isotope: 42: Electron shells: 2,8,18,5 : Electron configuration:. Minimum oxidation number-3 : Min. common oxidation no.-3 : Maximum oxidation number : 5 :. Arsenic is immediately dangerous to life or health at 5 mg m-3.. Arsenic (As) Arsenic is the 33rd element in the periodic table and has a symbol of As and atomic number of 33. It has an atomic weight of 74.92159 and a mass number of 75. Arsenic has thirty-three protons and forty-two neutrons in its nucleus, and thirty-three electrons in four shells. It is located in group fifteen, period four and block p of.

